Medicines regulatory bodies in the UK and Europe, the Medicines and Health products Regulatory Agency (MHRA) and European Medicines Agency (EMA) respectively, have released statements on the Oxford-AstraZeneca COVID-19 vaccine and cases of rare blood clots and low blood platelets.
On MHRA and EMA statements:
Dr Simon Clarke, Associate Professor in Cellular Microbiology at the University of Reading, said:
“After weeks of speculation, the MHRA and EMA have said that harmful blood clotting is likely a rare but significant side effect of the Oxford/AstraZeneca Covid-19 vaccine. Based on current available evidence, specific risk factors have not been confirmed, but the effect is significant enough for the JCVI to recommend that under 30s do not receive this vaccine.
“The main danger is that people don’t hear beyond there being an enhanced but small risk of harmful blood clotting in a small group of individuals and it puts those who are vulnerable to Covid-19 at risk.
“It would be very bad if people started questioning whether or not they should have their second dose of this safe and effective vaccine, which is essential for ensuring that their immunity to Covid-19 is as effective and long lasting as possible.
“This whole process has shown how important it is for the Government to be open and transparent about safety monitoring and not to gloss over or downplay any potential concerns there may be in the emerging data. This is especially important for those who were already sceptical in the first place about having the vaccine, so that there is trust in the public that their safety is at the heart of the vaccine roll out rather than being driven by a nationalistic push for a UK vaccine.
“The fact is that all vaccines carry risks. It’s good that all emerging risks are further investigated, but they need to be balanced against the risks of not being vaccinated. I would be happy to have an AstraZeneca jab, and while I will be more mindful of the symptoms of clots, I would gladly take the 1 in 250,000 chance of developing a blood clot, compared to having no protection against Covid-19.”
On MHRA and EMA statements:
Prof Andrew Pollard, Professor of Paediatric Infection and Immunity, said:
“Safety has been our priority throughout the development of the vaccine at University of Oxford in 2020, and we are reassured to see that safety monitoring continues under the close scrutiny of regulators and public health authorities as the vaccine is rolled out around the world.
“The identification of rare cases of blood clots, which might be associated with the vaccine, shows that the safety system works, and has also allowed MHRA and EMA to conclude that the benefits of vaccination continue to outweigh the risks, while putting in measures to help mitigate any possible risk.
“The pandemic coronavirus continues to be a huge threat to people throughout the world, and we continue with our mission to support global vaccination, not for profit, for the benefit of humanity.”
On MHRA statement and JCVI advice:
Prof Arne Akbar, President of the British Society for Immunology, said:
“As for all new vaccines and medications, the rollout of the new COVID-19 vaccines has been monitored extremely closely by the UK regulator, the Medicines and Healthcare products Regulatory Agency (MHRA). Today’s update highlights the importance of having this active monitoring in place and the fact that the regulator, working with the Joint Committee on Vaccination and Immunisation (JCVI), has acted to issue updated information today shows that this monitoring system is working well.
“After careful analysis, the JCVI have issued new guidance concerning who should receive the AstraZeneca/Oxford COVID-19 vaccine. This will have been the result of dozens of experts who have many years of experience in this area carefully scrutinising the available data. To be ultra cautious, they have advised that while the benefits of vaccination continue to outweigh the risks for everyone, people under 30 with no other health conditions should be offered an alternative vaccine to the AstraZeneca/Oxford vaccine where available.
“This is due to very rare reports of individuals developing blood clots with low platelet counts after receiving this vaccine. While the evidence is still accumulating to allow us to say definitively whether these symptoms are linked to the vaccine or not, the JCVI is taking a careful, proactive approach in acting now. The reason for this shift in emphasis for younger people is because the benefit to risk ratio is less pronounced than in older people who are at higher risk of severe COVID-19 disease. We still don’t have a clear understanding of what the mechanism behind these potential side-effects could be. Although there is some early evidence that there is an adverse immune response occurring that affects the platelets, we urgently need more research to clarify this.
“I would like to reassure people that the occurrence of these blood clots linked to low platelet counts after having the AstraZeneca/Oxford vaccine appears to be extremely rare – around one in 250,000. Vaccines are still the best way to protect yourself from catching COVID-19, which has a far higher risk of serious health effects. If you receive the call to get your COVID vaccine, I would urge you to do so. If you have any questions about vaccination, please speak to your GP or healthcare professional.”
On MHRA and EMA statements:
Prof Ian Douglas, Professor of Pharmacoepidemiology, London School of Hygiene & Tropical Medicine, said:
“The messages from both MHRA and EMA are in close agreement, which is reassuring. It’s also impressive to see the vaccine safety monitoring systems throughout Europe operating at such speed, with great rigour and transparency. The evidence from the case reports seems to be compelling enough to conclude at least a possible causal link between the AZ vaccine and rare clots accompanied by low platelet counts. Though I note the caution expressed here; it is still not proven beyond doubt. Two things do stand out beyond doubt. Firstly the well quantified and large benefits provided by all the currently available vaccines, and secondly that any risk from clotting appears to be very rare. The balance of risks and benefits shown by Prof Jonathan Van Tam for different age groups and in different levels of circulating virus helped demonstrate this to some degree, but we need to remember the risks from COVID are of course greater than admission to intensive care alone. Many people suffering consequences of COVID-19 such as clots and other long term problems won’t have been in intensive care but these are of course real and harmful side effects of COVID-19 and which need to be included in our assessment of the risks and benefits of vaccines.
“The move to recommend Pfizer or Moderna vaccines for under 30’s in the UK is a very cautious approach and makes some sense. Though we need to remember this doesn’t mean other vaccines are not also associated with rare clots, just that we have less evidence and experience with them at the moment.
“The reality of the incredible COVID vaccine role out speed and volume means we will continue to find out more about these rare clots and we may well see new safety signals with any of the vaccines. This is the nature of all effective medications; their rarer side effects only emerge when we use them at large scale.”
On MHRA statement:
Prof Martin Hibberd, Professor of Emerging Infectious Disease, London School of Hygiene & Tropical Medicine (LSHTM), said:
“I think the government has made a sensible choice in offering alternatives to people under 30, given the data. However, I would like to see the evidence that the other vaccines are safer. If they are, I would be tempted to increase the age where they can be recommended, to reduce people’s vaccine hesitancy. This should be possible in the UK, where two alternatives are already available.
“Also, I think the question on children is important. The AstraZeneca trials in children have been stopped, while Pfizer continue – this should be based on evidence that the data in the 18–30-year-olds looks different between AstraZeneca and Pfizer – which may need to come from countries outside of the UK, as relatively few people of this age have been vaccinated within the UK.
“Fully opening-up society is best done when more than 85% of the population are vaccinated, to gain from both reduced transmission as well as reduced disease. To achieve this, all age groups should benefit from the vaccines.”
On MHRA and EMA statements:
Prof Kevin McConway, Emeritus Professor of Applied Statistics, The Open University, said:
“I don’t disagree with the JVCI recommendation, but I do want to draw attention to a couple of aspects of the evidence. First, there’s still a great deal of uncertainty about all this, and though that did come across in a lot of what was said in the UK press conference and also the EMA press conference, I think it bears repeating. As an example, one thing that needs to be done in checking whether a possible vaccine adverse effect is actually caused by the vaccine is to compare how often the effect happened after vaccination compared to how often it happens in health unvaccinated people. Just to point out how difficult this can be, a recent EMA report about blood clots, https://www.ema.europa.eu/en/documents/prac-recommendation/signal-assessment-report-embolic-thrombotic-events-smq-covid-19-vaccine-chadox1-s-recombinant-covid_en.pdf, looked at the background risk of CVST, one of the serious blood clotting disorders under consideration (pages 6-7). They say that, in a million people followed up for a year (before Covid), somewhere between 2 and 5 might have a CVST, but then point out that more recent studies say it might be 13 or 16 cases, not 2 or 5, and that this seems to be because data are being collected more carefully. With this uncertainty about what would happen without Covid and without vaccination, it’s difficult for the regulatory authorities to make the comparisons they need to make. And when they do, there could still be considerable uncertainty involved, as was stated.
“I realise that what’s being discussed is particular types of CVST also involving low platelets (thrombocytopenia), and I’m not questioning the statements that a link with the AZ vaccine looks plausible and possibly quite likely. But all this is, inevitably, based on observational data to a considerable extent. In other research contexts I’d be warning you about not drawing causal conclusions from observational data. What the various authorities have done already goes well beyond just looking at observational data, which is why they can say what they did say about possible cause. But the observational nature of the reports of adverse effects, and the statistical uncertainty because the adverse events are so rare, is why they can’t yet be certain what’s going on. More research is being done and, eventually, we’ll know more precisely.
“I think all that needs to be borne in mind when thinking about the slides from the Winton Centre that Prof Jonathan Van Tam presented. They do get across well the balance between risks and benefits of vaccination – but maybe they get it across too well, in that they risk making us think we know things more precisely than we really do. We already saw, because there were three different slides for different infection risk levels, that the balance depends on the risk of infection, and we don’t know how that will change in coming weeks and months. But is the rate of ICU admissions over 16 weeks the right measure of the benefit from vaccination? Actually there isn’t a single ‘right’ measure – there’s a lot to try to balance out. And what’s the statistical (and other) uncertainty in those figures, and in the figures on the risk of serious harms from the vaccine? Is the risk of serious harms really exactly 5.5 times as big for people aged 20-29 as people aged 60-69, as the slides indicated? Unlikely – that might be the best current estimate, but it’s surely subject to a lot of statistical uncertainty, and also to the fact that the rate at which people have, so far, reported these adverse effects is unlikely to be a totally accurate measure of the true rate at which the events occur (the incidence rate).
“I’m absolutely not running down the work of the various regulators and decision makers, or indeed of the statisticians who did an excellent job in producing those slides. I just want to warn against anyone thinking this can be the last word on the matter. There’s still so much that we don’t know, and can’t know yet. Evidence, understanding, and policies will change as we learn more.”
On MHRA statement:
Prof Lawrence Young, Professor of Molecular Oncology, Warwick Medical School, said:
“Evidence is firming up that the AZ/Oxford jab is associated with a very rare side effect of blood clots associated with low levels of platelets. But the benefits of the vaccine far outweigh this risk. The low number of cases of this side effect make it difficult to identify trends but there is a suggestion for slightly higher incidence in the younger adults – hence the advice that those under 30 should be offered an alternative COVID-19 vaccine. Rare side effects can only be seen when a vaccine is used at scale but we still need more data to firm up the association of the AZ/Oxford vaccine with these rare blood clots. This will come from keeping a watchful eye on reported side effects as more people are vaccinated. If the association is confirmed then possible risk factors could be identified. One suggestion is that these blood clots are associated with an aberrant immune response that targets platelets – the cells in our blood responsible for blood clotting. As increased blood clotting is a common feature of COVID-19, it could be that this effect is due to people getting infected with the virus soon after vaccination or it could be a direct effect of this particular type of vaccine. The AZ/Oxford vaccine is based on similar technology (recombinant adenoviruses) to that used in other vaccines (e.g. the Johnson & Johnson and Sputnik V vaccines) so it will be important to check whether these vaccines are also associated with the same side effects.”
On MHRA and EMA statements:
Prof Saad Shakir, Director of the DSRU, said:
“Both the European and UK briefings acknowledged evidence that these very rare blood clots are possibly caused by the AstraZeneca COVID-19 vaccine, and both regulators talked about how serious a threat COVID-19 remains for us.
“The main difference was that in Europe they said the blood clots affected all ages and both genders, while in the UK, they talked about a different risk of these rare blood clots in younger people. The UK recommendation that under 30s receive an alternative vaccine is a welcome and suitably cautious move.
“I appreciate this news will cause some people concern. I would reiterate that these events are very rare and I hope people can take some comfort from the knowledge that robust, meticulous research and monitoring is on-going into any possible side effects.”
On MHRA statement:
Prof Sheila Bird, formerly Programme Leader, MRC Biostatistics Unit, University of Cambridge, said:
“Vaccine-type matters when considering serious adverse events; also, vaccine effectiveness. Correct at-risk denominators matter (eg by age-group and gender) for the number of persons to whom one or more doses was administered per vaccine-type. Today’s MHRA briefing did not provide at-risk denominators by age-group for 1st dose recipients of the Oxford/AstraZeneca (OxAZ) prior to 31 March 2021.
“Today’s briefing announced 79 case reports of cerebral venous sinus (or other**) thrombosis (CVST) with low platelet count which occurred after a 1st dose of AstraZeneca was given to 18 million persons. Fifty-one of these serious adverse events occurred in women. There were 19 deaths.
“The briefing included a key risk-benefit comparison by age-group per 1 million OxAZ-vaccinees, see Table. But yellowcard reports by age-group and at-risk denominators were not disclosed. Potential benefit depends, of course, on what has been assumed about vaccine effectiveness for preventing COVID-19 disease severe enough to require admission to intensive care. Potential benefit is itself vaccine-specific but, in scenario modelling for SAGE, has generally been assumed to be lower for Oxford/AstraZeneca vaccine than for Pfizer/BioNTech.
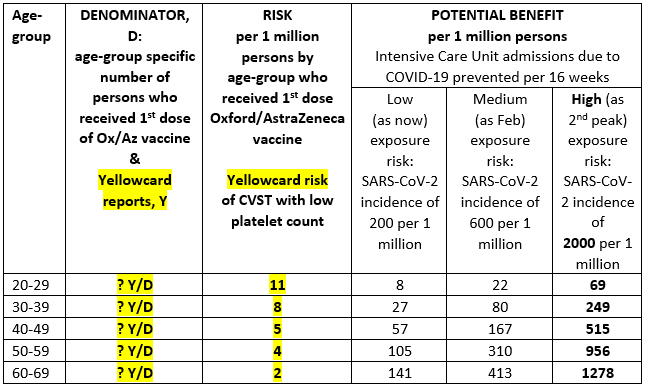
“Under-reporting means that UK’s yellowcard-reported-risk underestimates actual-risk. However, the UK’s course-correction – made for persons under 30 years rather than under 40 years – suggests confidence on the part of MHRA that yellow-card-reporting for COVID-vaccines about this rare serious adverse event is substantially better than 10% of actual-risk.
**44 of the 79 cases were of CVST with thrombocytopenia; 35 of the 79 cases were of thrombosis in other major veins with thrombocytopenia.”
On MHRA statement:
Prof Anthony Harnden, Deputy Chair of JCVI, said:
“The UK vaccination programme has saved at least 6000 lives in the first three months and will help pave the way back to normal life for us all. More than 20 million doses of the Oxford AZ vaccine have been given to date. But there have been reports of extremely rare events of blood clotting associated with low platelets in a small number of people following the first dose of Oxford AZ vaccine. Whilst the mechanism for this is uncertain there does seem to be a trend for increasing incidence of this event with decreasing age. And although the chance of any person receiving the vaccine experiencing a blood clot with low platelets is extremely small, because the risk of severe Covid in the under 30s with no underlying illness is also small JCVI feel as a precautionary measure it is appropriate for those in this age group to be offered an alternative Covid vaccine when their turn comes for their first dose of a vaccine. JCVI will continue to further monitor the safety data as it emerges. What is clear it that for the vast majority of people the benefits of the Oxford AZ vaccine far outweigh any extremely small risk and the Oxford AZ vaccine will continue to save many from suffering the devastating effects that can result from a Covid infection.”
On MHRA and EMA statements:
Prof David Werring, Professor of Clinical Neurology, UCL Institute of Neurology, UCL, said:
“The EMA and MHRA statements are helpful in providing further clarity about the very rare blood clots with low blood platelet counts – mainly in the brain (cerebral venous sinus thrombosis) but also in other parts of the body – that have occurred after the AstraZeneca vaccine.
“Both the EMA and MHRA emphasise that the overall benefit of the vaccine in preventing illness, hospitalisation and death from COVID-19 clearly outweighs the risks of this rare potential complication; thus, vaccination programs should continue as planned. However, the latest UK data indicate that the potential benefits most clearly outweigh serious harms in older people (over the age of 40), particularly with increasing COVID-19 exposure risk; in younger people aged 18-29 the risks are more finely balanced (though still in favour of vaccination in most situations) leading to the suggestion of these individuals being offered an alternative to the AstraZeneca vaccine as a precaution.
“We still urgently need more research to first understand which individuals are at highest risk of these unusual blood clots in the brain, which although very rare, can often be serious or fatal. Although most cases were in females under the age of 60, this varied across countries with different vaccination strategies so did not allow specific risk factors (such as age or gender) to be identified. Second, the specific blood test findings (low blood platelets, associated specific antibodies, and increased blood clotting) suggest that an immune reaction could be an important mechanism of post-vaccine thrombosis, but being sure about a definite causal link needs much more scientific work and confirmation.
“The recognition that this unusual and distinct pattern of thrombosis with low platelets should now be considered a possible adverse drug reaction, is helpful and emphasises the need for ongoing careful and systematic surveillance. While vaccination programs should continue, a key message is that people receiving the vaccine, their doctors, and other healthcare professional need to be aware of the possible symptoms of thrombosis (especially in the brain), including: a severe headache that is sudden or progressive; neurological symptoms (for example face, arm or leg weakness, disturbance of speech or vision, confusion, drowsiness or seizures); shortness of breath; or leg pain and swelling. Specific treatments in hospital may improve the outcome from cerebral venous sinus thrombosis – but only if given soon enough – so it is crucial that people seek medical attention urgently if they experience these symptoms between 4 and 21 days after vaccination.”
On MHRA and EMA statements:
Dr Michael Head, Senior Research Fellow in Global Health, University of Southampton, said:
“We have seen an update from the UK and EU regulators, suggesting that these thrombotic events may have been a causal, but rare, adverse event from the Oxford AstraZeneca vaccine. This link is still not proven, but is now thought to be increasingly plausible. From the EMA statement, there were 86 events investigated across 25 million doses administered. This signal equates to approximately 1 possible adverse event in every 290 000 doses.
“Revised recommendations from the UK regulators suggest that younger adults aged 18-29 should ideally receive an alternative COVID-19 vaccine, such as Pfizer or Moderna.
“It’s important to emphasise that adverse events happen with all medicines, and vaccines are no exceptions. Safety surveillance is vital in picking up and assessing signals that emerge from the data. There were some cases of severe anaphylaxis with the Pfizer vaccine early in the UK rollout. These were openly investigated, guidance subsequently updated, and the rollout continued with high public confidence. Hopefully, we will see similar outcomes here in the UK with the Oxford AstraZeneca product, and also that European countries can get their vaccine administrations back on track. The harm from withdrawing the vaccine altogether is almost certainly going to be much greater than the harm from rare adverse events.
“The Oxford AstraZeneca vaccine is a vital tool in the global strategy to contain the pandemic. It is being manufactured in large numbers, is stored at refrigeration temperatures and thus easier to transport, cost per dose is cheap, and key to the COVAX distribution to low- and lower-middle income countries. Maintaining public confidence is so important. An open transparent process to assessing safety concerns must be part of that.”
EMA statement: https://www.ema.europa.eu/en/news/astrazenecas-covid-19-vaccine-ema-finds-possible-link-very-rare-cases-unusual-blood-clots-low-blood
SMC Q&A Briefing with vaccine safety and thrombosis experts.
All our previous output on this subject can be seen at this weblink:
www.sciencemediacentre.org/tag/covid-19
Declared interests
Prof Andrew Pollard: “Andrew Pollard is chair of the UK Department of Health and Social Care’s (DHSC) Joint Committee on Vaccination and Immunisation (JCVI), but does not chair or participate in the JCVI coronavirus committee, and is a member of the World Health Organization’s (WHO) Strategic Advisory Group of Experts. He has received research funding for coronavirus vaccine research from UKRI, CEPI and NIHR. Oxford University has entered into a partnership with Astra Zeneca for the development of a coronavirus vaccine.”
Prof Martin Hibberd: “No COI to declare.”
Prof Kevin McConway: “I am a Trustee of the SMC and a member of its Advisory Committee. I am also a member of the Public Data Advisory Group, which provides expert advice to the Cabinet Office on aspects of public understanding of data during the pandemic. My quote above is in my capacity as an independent professional statistician.”
Prof Saad Shakir: “Saad Shakir is an employee of the Drug Safety Research Unit (DSRU), an independent charity (No. 327206), which works in association with the University of Portsmouth.
The DSRU is conducting a safety study of the AstraZeneca vaccine, proactively questioning 10,000 vaccinees about possible side effects. The study is in its early stages and the DSRU will independently publish any findings.
Prof Shakir is a member of the Data Safety Monitoring Boards for Diurnal and Biogen.”
Prof Anthony Harnden: “I am Deputy Chair of JCVI”
None others received.